Abstract
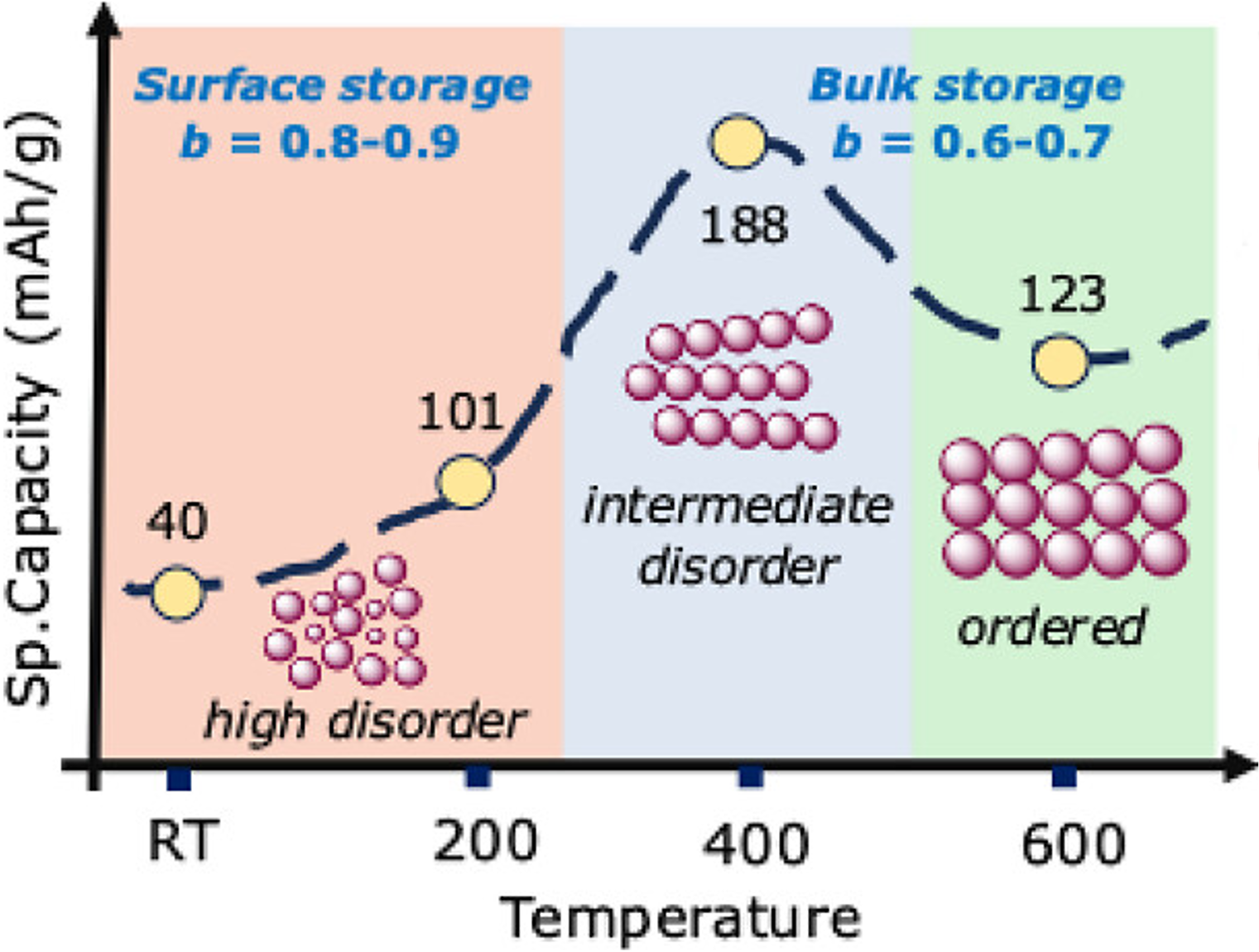
The inability of graphite to store Na+ ions at low potentials necessitates the use of disordered materials as anodes in Na-ion batteries, wherein complex degradative pathways limit cycle life. This study delineates the influence of synthetically induced material disorder in facilitating stable, high-rate sodium ion storage in a series of anatase titania analogs. The degree of disorder in the titanium oxyhydroxide (TiOx(OH)y-Tanneal) materials, synthesized via a simple, solution-phase sol–gel protocol employing a Ti(III) precursor, is systematically varied by tuning the postsynthesis annealing temperature (Tanneal). The variable disorder in TiOx(OH)y-Tanneal stems from the distribution of residual Ti(III), N-dopants, and O-vacancies in these materials as a function of Tanneal, as mapped by XPS, bulk-sensitive resonance inelastic X-ray scattering (RIXS), and EPR spectroscopy. In terms of electrochemical performance, we observe a unique, nonmonotonic dependence of Na+ storage capacity on the synthetically induced degree of disorder in TiOx(OH)y-Tanneal, instead of a simple inverse size dependence, as normally seen in the case of fully crystalline anatase materials. Further in contrast to crystalline anatase, the disorder in TiOx(OH)y-Tanneal opens up a bulk mode of Na+ storage instead of a purely surface confined or capacitive mode, as determined by scan rate-dependent cyclic voltammetry. TiOx(OH)y-400, with an intermediary amount of disorder in the prepared series of materials, shows the highest specific capacity (∼188 mAh/g) and cycling stability for Na+ storage at elevated rates. These results are in agreement with our first-principles simulated data exhibiting a higher number of possible sites that Na+ can occupy at an intermediate degree of disorder in a TiOx(OH)y structure.