Abstract
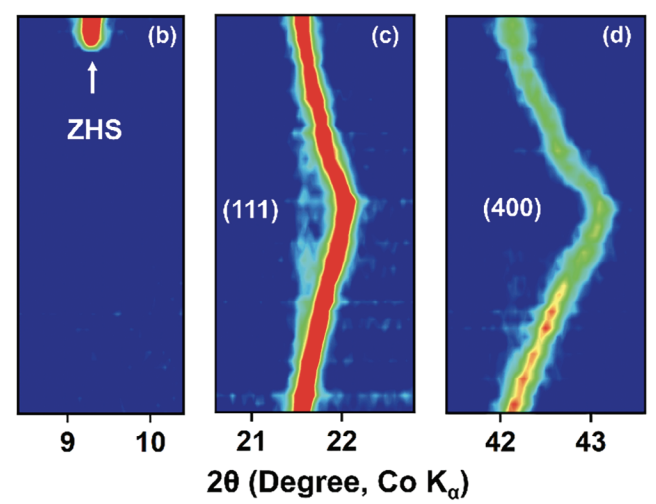
H+ co-intercalation chemistry of the cathode is perceived to have damaging consequences on the low-rate and long-term cycling of aqueous zinc batteries, which is a critical hindrance to their promise for stationary storage applications. Herein, the thermodynamically competitive H+ storage chemistry of an attractive high-voltage cathode LiMn2O4 is revealed by employing operando and ex-situ analytical techniques together with density functional theory-based calculations. The H+ electrochemistry leads to the previously unforeseen voltage decay with cycling, impacting the available energy density, particularly at lower currents. Based on an in-depth investigation of the effect of the Li+ to Zn2+ ratio in the electrolyte on the charge storage mechanism, a purely aqueous and low-salt concentration electrolyte with a tuned Li+/Zn2+ ratio is introduced to subdue the H+-mediated charge storage kinetically, resulting in a stable voltage output and improved cycling stability at both low and high cathode loadings. Synchrotron X-ray diffraction analysis reveals that repeated H+ intercalation triggers an irreversible phase transformation leading to voltage decay, which is averted by shutting down H+ storage. These findings unveiling the origin and impact of the deleterious H+-storage, coupled with the practical strategy for its inhibition, will inspire further work toward this under-explored realm of aqueous battery chemistry.